Introduction
For healthcare professionals working in any clinical environment, filter solution represents foundational knowledge. This guide provides a comprehensive overview of key concepts, clinical applications, and best practices.
Background and Clinical Context
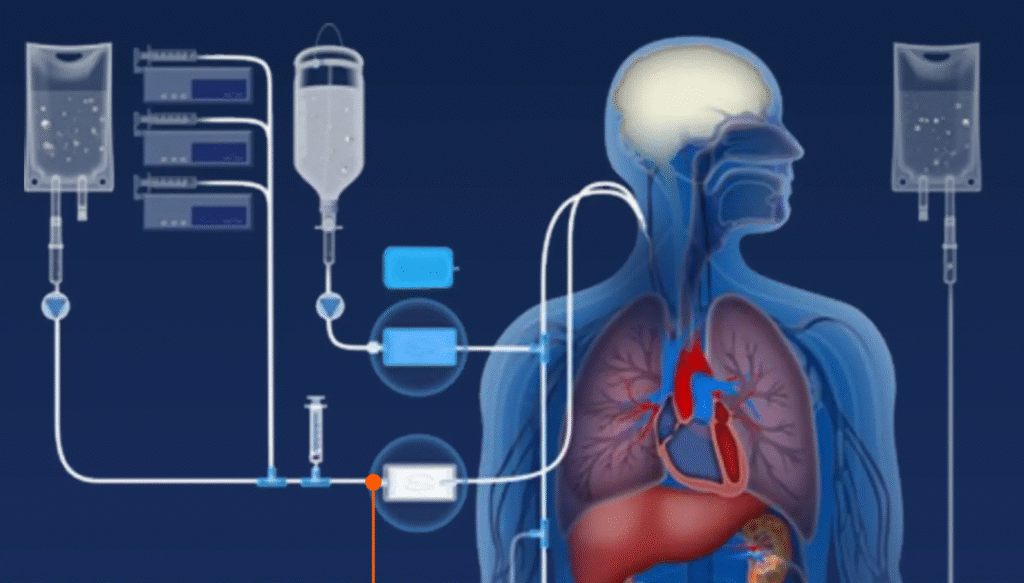
The infusion set bridges the gap between a fluid container and the patient’s circulatory system. It ensures accurate, controlled, and sterile delivery of intravenous solutions. Medical-grade materials and precision manufacturing are essential to its safe performance in clinical environments.
Core Components of an Infusion Set
A standard infusion set consists of a piercing spike, drip chamber with integrated filter, flexible PVC tubing, a roller clamp, a Y-site injection port, and a luer connector at the distal end. Each component must meet strict regulatory standards to ensure patient safety.
How Infusion Rate Is Calculated
The infusion rate depends on the prescribed fluid volume and required duration. Clinicians use the drip factor and the formula: drops per minute equals total volume in mL multiplied by the drip factor, divided by total time in minutes. Accurate calculation prevents under-infusion and over-infusion.
How to Use This Knowledge Effectively
Modern clinical practice uses several specialized infusion sets. Burette sets hold a measured volume in a calibrated chamber, ideal for pediatric use. Photosensitive sets block UV light to protect medications like nitroprusside. Pump-specific sets are designed for volumetric infusion pump compatibility.
- Confirm patient identification before beginning any infusion
- Verify the five rights of medication administration at every step
- Check IV bag and tubing for particulates, cloudiness, or leakage
- Maintain clear documentation of all IV therapy in the patient record
- Reassess the IV site and patient response to therapy regularly
Evidence-Based Recommendations
Quality assurance involves multiple checks including raw material testing, in-process quality control, and final product validation. ISO 8536 standards govern the design and testing of infusion equipment. Hospitals should source sets only from certified, reputable manufacturers.
Frequently Asked Questions
FAQ 1: What should I do if I notice a defect in IV equipment?
Any defective IV equipment must be removed from service immediately. Document the defect, report to your supervisor, complete an incident or adverse event report, and retain the defective product for investigation if required by institutional policy.
FAQ 2: How does patient age affect equipment selection?
Patient age significantly influences equipment selection. Neonates and infants require micro-drip sets and smaller cannulas. Pediatric patients need smaller volumes with more precise control. Adults typically use standard macro-drip sets for routine fluid therapy.
FAQ 3: Are there environmental considerations for disposing of IV equipment?
Used IV equipment including tubing, needles, and filters must be disposed of as clinical waste following local regulations. Sharp components go into approved sharps containers. Tubing and non-sharp parts go into clinical waste bins.
Conclusion
Whether you are new to clinical practice or a seasoned professional, staying current on filter solution is vital. The concepts outlined here contribute directly to safer patient care and more effective medical treatment every day.


